Compliance Requirements We Design Against
Our templates are built to align with the major labeling frameworks your teams work under, then customized with your SDS, IFU and branding.
OSHA Hazard Communication & GHS
OSHA’s Hazard Communication Standard (HazCom 2012) requires chemical manufacturers and importers to label containers with harmonized elements: product identifier, supplier information, signal word, hazard statements, precautionary statements and GHS pictograms.
- Primary container labels tied directly to the SDS.
- Workplace / secondary container labels consistent with training.
- Posters and quick-reference cards to decode pictograms.

BS5609 for Marine & Export
BS5609 Part 2 and 3 specify strict performance tests for label materials and printed images used on marine chemical drums—salt spray, abrasion and three-month sea immersion.
- Certified media + ink combinations where required by carriers.
- Layouts that combine GHS, UN number and transport markings.

Healthcare, CLIA & Device Expectations
Labs and device manufacturers must ensure labels stay legible across storage, processing and transport—often under CLIA, FDA and international expectations. We help align bench-level labels with your device instructions for use and submission packages.

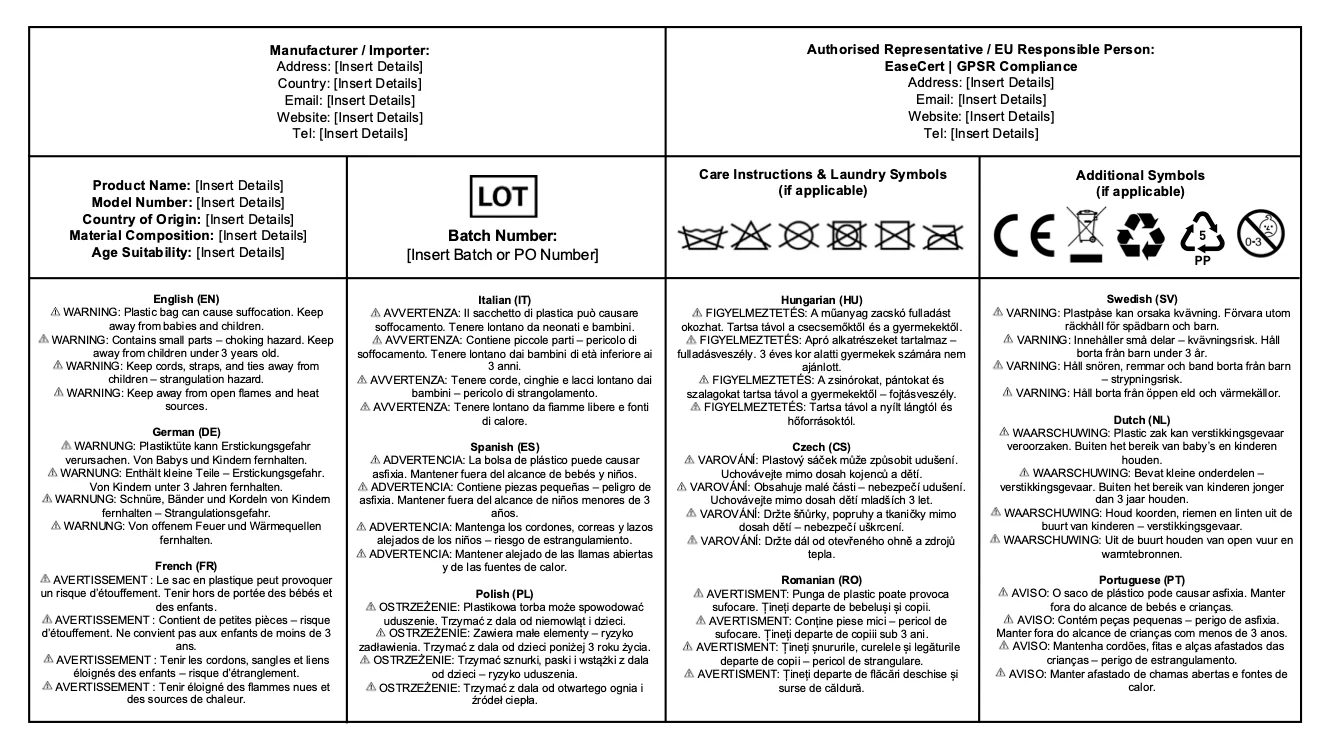
GPSR-Style Product Safety Information
The EU’s General Product Safety Regulation (GPSR) strengthens requirements for clear, traceable consumer product information. Our label designs can incorporate product identifiers, safety warnings and traceability fields so that your packaging is GPSR-ready.
FDA LDT / IVD Transition
For laboratories running LDTs that are transitioning toward IVD-style regulation, we can align your labels with your evolving QMS and device files.
See the dedicated FDA page for more on timelines and how label content fits into your broader quality system:
